21 CFR Part 11 compliant system in Western Blots and 1D Gels Analysis
Fully Part 11 compliant system for Blot and 1D Gel analysis. Our answer to data integrity.

Full 1D analysis system validation with new extension LabImage PQ
The new PQ extension performs full system validation with a generic 1D gel

Batch Processing for 1D Gels and Blots with predefined methodes
Analyze batches of gel and blot images in seconds and create your own methodes
Latest News
Kapelan Bio-Imaging Successfully Certified to ISO 13485: Quality Management Meets Legal Requirements, Qualifies Kapelan Bio-Imaging as Provider of Medical Device and IVD Software
Leipzig, Germany, 240313: Kapelan Bio-Imaging is pleased to announce that the company has successfully passed the surveillance audit for ISO 13485. This certification confirms that Kapelan Bio-Imaging’s quality management…
Kapelan Bio-Imaging offers new calibration service for grayscale charts
Kapelan Bio-Imaging, a provider of innovative solutions for biomedical image analysis, proudly introduces its new grayscale chart calibration service. Customers can now have their reflected and transmitted light charts precisely…
New PQ Extension: Ensuring Accurate Analysis of 1D Gels through Full System Validation
The PQ Extension is an advanced software tool by Kapelan Bio-Imaging that extends the capabilities of LabImage 1D. It enables users to run a full analysis system validation for any…
New 21 CFR Part 11 System
Coming within the next releases of LabImage Core we offer a brand new 21 CFR Part 11 system. It offers a major breakthrough towards flexibility and adaptation. Almost every feature of the LabImage Apps can be controlled using the new right management and grouped into specific roles. Roles can be bent to user groups. Moreover, each of needed tools as macros, mw standards and test definitions can be versioned now allowing the usage of a release process.
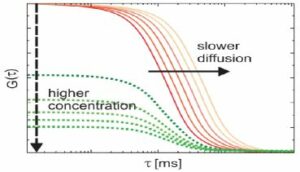
Fluorescence correlation spectroscopy (FCS)
FCS is a statistical analysis, via time correlation, of stationary fluctuations of the fluorescence intensity. The analysis provides kinetic parameters of the physical processes underlying the fluctuations. We are part of a project team to develop and commercialize a new FCS device and software. Our part is to develop device controlling workflows as well as data analysis.
Digital Holographic Microscopy (DHM)
Digital holographic microscopy (DHM) is digital holography applied to microscopy. Digital holographic microscopy distinguishes itself from other microscopy methods by not recording the projected image of the object. Instead, the light wave front information originating from the object is digitally recorded as a hologram, from which a computer calculates the object image by using a numerical reconstruction algorithm. We are involved into the development of a new device and software to bring DHM into new fields of application.
Full 1D system validation – PQ extension
The PQ Extension is an advanced software tool that extends the capabilities of LabImage 1D. It enables users to run a full analysis system validation for any 1D gel or western blot, providing proof of analysis quality. This is a significant breakthrough as it allows users to ensure that their analysis results are accurate and reliable. It is the first tool worldwide to offer a full system validation in 1D analysis. More
Latest Projects
LabImage Applications
Latest Scientific Publications using LabImage
Paper: ROCK1/2 signaling contributes to corticosteroid-refractory acute graft-versus-host disease
Abstract Patients with corticosteroid-refractory acute graft-versus-host disease (aGVHD) have a low one-year survival rate. Identification and validation of novel…
Paper: Hepatoprotective effect of Physalis divaricata in paracetamol induced hepatotoxicity: In vitro, in silico and in vivo analysis
Abstract Ethnopharmacological relevance Physalis divaricata D. Don. is an erect weed of family Solanaceae. The root extract of this plant…
Paper: Proteins in human body fluids contain in vivo antigen analog of the melibiose-derived glycation product: MAGE
Abstract Melibiose-derived AGE (MAGE) is an advanced glycation end-product formed in vitro in anhydrous conditions on proteins and protein-free amino…
About Us.
Kapelan Bio-Imaging
Kapelan Bio-Imaging is a bio-imaging company from Leipzig, Germany. We develop and distribute software and imager for digital image analysis in research, routine and pharma.