


Komplette Systemvalidierung mit neuem generischen 1D Gel
Neue Erweiterung LabImage PQ liefert komplette Systemvalidierung mit kalibriertem Gel
Aktuelles
ISO 13485-Rezertifizierungsaudit erfolgreich bestanden
Wir freuen uns, bekannt geben zu können, dass wir unser ISO 13485-Rezertifizierungsaudit erfolgreich bestanden haben! Dieser Erfolg unterstreicht unser kontinuierliches Engagement für höchste Standards im Qualitätsmanagement für Medizinproduktesoftware. Vielen Dank…
Startschuss für das Netzwerk für Therapeutische Nukleinsäuren
Fachexperten aus Wirtschaft und Wissenschaft bündeln ihre Kompetenzen, um Lösungen aus dem Entwicklungslabor in die Klinik zu überführen. Therapeutische Nukleinsäuren bzw. Oligonukleotide haben ein enormes Potential für die Behandlung derzeit…
Kick-Off Meeting für Projekt zum Thema Fluoreszenz-Korrelations-Spektroskopie
Am 21.1.2025 fand in Jena das Kick-Off Meeting für ein neues Projekt statt zum Thema Fluoreszenz-Korrelations-Spektroskopie – ein sperriges Wort aber eine vielversprechende Technologie, zu der wir neue KI Methoden…
Komplette Überarbeitung des 21 CFR Part 11 Systems
 Mit den nächsten Versionen von LabImage Core bieten wir ein komplett neues 21 CFR Part 11-System an. Es stellt einen Durchbruch in Richtung Flexibilität und Anpassbarkeit dar. Nahezu alle Funktionen der LabImage Apps können über die neue Rechteverwaltung gesteuert und in bestimmte Rollen gruppiert werden. Rollen können auf Benutzergruppen zugeschnitten werden. Darüber hinaus können jetzt alle benötigten Tools wie Makros, MW-Standards und Testdefinitionen versioniert werden, was die Verwendung eines Release-Prozesses ermöglicht.
Mit den nächsten Versionen von LabImage Core bieten wir ein komplett neues 21 CFR Part 11-System an. Es stellt einen Durchbruch in Richtung Flexibilität und Anpassbarkeit dar. Nahezu alle Funktionen der LabImage Apps können über die neue Rechteverwaltung gesteuert und in bestimmte Rollen gruppiert werden. Rollen können auf Benutzergruppen zugeschnitten werden. Darüber hinaus können jetzt alle benötigten Tools wie Makros, MW-Standards und Testdefinitionen versioniert werden, was die Verwendung eines Release-Prozesses ermöglicht.
Fluoreszenzkorrelationsspektroskopie (FCS)
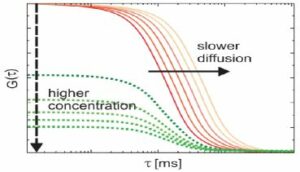 Bei der Fluoreszenzkorrelationsspektroskopie (FCS) handelt es sich um eine statistische Analyse stationärer Schwankungen der Fluoreszenzintensität mittels Zeitkorrelation. Die Analyse liefert kinetische Parameter der physikalischen Prozesse, die den Schwankungen zugrunde liegen. Wir sind Teil eines Projektteams zur Entwicklung und Vermarktung eines neuen FCS-Geräts und einer neuen Software. Unsere Aufgabe besteht darin, Arbeitsabläufe zur Gerätesteuerung sowie Datenanalyse zu entwickeln.
Bei der Fluoreszenzkorrelationsspektroskopie (FCS) handelt es sich um eine statistische Analyse stationärer Schwankungen der Fluoreszenzintensität mittels Zeitkorrelation. Die Analyse liefert kinetische Parameter der physikalischen Prozesse, die den Schwankungen zugrunde liegen. Wir sind Teil eines Projektteams zur Entwicklung und Vermarktung eines neuen FCS-Geräts und einer neuen Software. Unsere Aufgabe besteht darin, Arbeitsabläufe zur Gerätesteuerung sowie Datenanalyse zu entwickeln.
Digitale holographische Mikroskopie (DHM)
 Digitale holographische Mikroskopie (DHM) ist eine auf die Mikroskopie angewendete digitale Holographie. Die digitale holographische Mikroskopie unterscheidet sich von anderen Mikroskopiemethoden dadurch, dass sie nicht das projizierte Bild des Objekts aufzeichnet. Stattdessen wird die vom Objekt ausgehende Lichtwellenfrontinformation digital als Hologramm aufgezeichnet, aus der ein Computer mithilfe eines numerischen Rekonstruktionsalgorithmus das Objektbild berechnet. Wir sind an der Entwicklung eines neuen Geräts und einer neuen Software beteiligt, um DHM in neue Anwendungsfelder zu bringen.
Digitale holographische Mikroskopie (DHM) ist eine auf die Mikroskopie angewendete digitale Holographie. Die digitale holographische Mikroskopie unterscheidet sich von anderen Mikroskopiemethoden dadurch, dass sie nicht das projizierte Bild des Objekts aufzeichnet. Stattdessen wird die vom Objekt ausgehende Lichtwellenfrontinformation digital als Hologramm aufgezeichnet, aus der ein Computer mithilfe eines numerischen Rekonstruktionsalgorithmus das Objektbild berechnet. Wir sind an der Entwicklung eines neuen Geräts und einer neuen Software beteiligt, um DHM in neue Anwendungsfelder zu bringen.
Vollständige Validierung 1D System mit PQ
Die PQ Exte nsion ist ein neues Softwaretool, das die Funktionen von LabImage 1D erweitert. Benutzer können damit eine vollständige Validierung des Analysesystems für jedes 1D-Gel oder jeden Western Blot durchführen und so die Qualität der Analyse nachweisen. Dies ist ein bedeutender Durchbruch, da Benutzer so sicherstellen können, dass ihre Analyseergebnisse genau und zuverlässig sind. Es ist das erste Tool weltweit, das eine vollständige Systemvalidierung in der 1D-Analyse bietet. Mehr
nsion ist ein neues Softwaretool, das die Funktionen von LabImage 1D erweitert. Benutzer können damit eine vollständige Validierung des Analysesystems für jedes 1D-Gel oder jeden Western Blot durchführen und so die Qualität der Analyse nachweisen. Dies ist ein bedeutender Durchbruch, da Benutzer so sicherstellen können, dass ihre Analyseergebnisse genau und zuverlässig sind. Es ist das erste Tool weltweit, das eine vollständige Systemvalidierung in der 1D-Analyse bietet. Mehr
Aktuelle Projekte
Aktuelle Publikationen mit LabImage
Paper: Hepatoprotective effect of Physalis divaricata in paracetamol induced hepatotoxicity: In vitro, in silico and in vivo analysis
Abstract Ethnopharmacological relevance Physalis divaricata D. Don. is an erect weed of family Solanaceae. The root extract of this plant…
Paper: Proteins in human body fluids contain in vivo antigen analog of the melibiose-derived glycation product: MAGE
Abstract Melibiose-derived AGE (MAGE) is an advanced glycation end-product formed in vitro in anhydrous conditions on proteins and protein-free amino…
Paper: Omeprazole prevents stress induced gastric ulcer by direct inhibition of MMP-2/TIMP-3 interactions
Abstract The healing of damaged tissues in gastric tract starts with the extracellular matrix (ECM) remodeling by the action of…
LabImage Applikationen

Über uns.
Kapelan Bio-Imaging
Kapelan Bio-Imaging ist ein Bio-Imaging-Unternehmen aus Leipzig, Deutschland. Wir entwickeln und vertreiben Software und Imager für die digitale Bildanalyse in Forschung, Routine und Pharma.










