History
The history of Kapelan
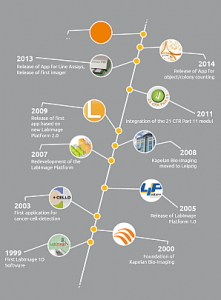
As spin-off from a German diagnostics specialist in cancer prevention, Kapelan Bio-Imaging launched the first software product for 1D gel analysis in 1999. In the next few years, the company developed several additional bioinformatics solutions, including software for fluorescence imaging and blot analysis.
After Kapelan Bio-Imaging successfully introduced the LabImage platform in 2005, the software was subsequently extended to better serve the needs of research institutes as well as the pharmaceutical and diagnostics industry. In the last ten years, thousands of customers worldwide have enjoyed Kapelan’s computer-based image analysis solutions and the LabImage platform has evolved to cover the full range of bio imaging software.
As a spin-off Kapelan founded a New Media company which was part of Kapelan up to January 2010. Since that Kapelan was renamed to Kapelan Bio-Imaging to strongly focus on Bio-Imaging solutions. In January 2010 the headquarter was moved to Leipzig/Germany.
In December 2010 Kapelan celebrated the 10 years anniversary followed by the release of the new LabImage 1D in January 2011.
With a worldwide outreach and a fast-growing network of partners and resellers, Kapelan Bio-Imaging boasts a strong integration into the German as well as the international research community and is ideally positioned to become a market leader in bio-imaging.
In 2013 Kapelan released the LabImage Application Pool which brings a maximum of flexibility to the imaging community. Specific imaging application on your finger tips is the main focus of the LabImage Platform 3.0. More and more clients in research and diagnostics choose the LabImage Platform as a flexible and extandable software enviroment. In combination with the new LabImager device series custom specific analysis systems find their way to the labs worldwide.
Within following years the part 11 system was rebuilt so many more pharma companies switched to LabImage 1D.
In 2022 Kapelan integrated quality management system according to ISO 13485 and maintained it over the years.
In the last years Kapelan was requested to cooperate in many research projects to bring in experience in imaging.
Also we started to create a fully new part 11 system with much more flexibilty.
Starting in 2024 Kapelan developed a cross plattform solution and worked on LabImage Cloud service. This will bring imaging to desktop, browser and app.
Kapelan celebrated its 25th anniversary in 2025.
Since that time several new applications started as well as our own device production and integration.
Founded: 2000
Number of Employes: 5
Type: private held limited
Competence: image analysis, high throuput processing, custom software development, development of image readers
Short Overview